Get your business and economy news from the world
Provided by AGPAlpha-1 Antitrypsin Deficiency Market Set for Significant Growth Driven by Rising Demand | Persistence Market Research
The Alpha-1 Antitrypsin Deficiency market is growing globally, driven by genetic respiratory disorders, advanced therapies, diagnostics, and digital health.
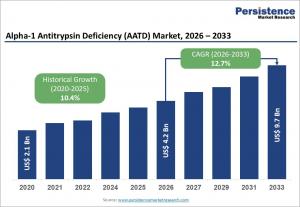
LONDON, UNITED KINGDOM, May 6, 2026 /EINPresswire.com/ -- The global alpha-1 antitrypsin deficiency (AATD) market is projected to reach a valuation of US$ 4.2 billion in 2026 and expand to US$ 9.7 billion by 2033, reflecting a robust compound annual growth rate (CAGR) of 12.7% during the forecast period of 2026–2033. The market’s growth is primarily driven by the increasing prevalence of genetic respiratory disorders, such as chronic obstructive pulmonary disease (COPD) and emphysema, which are frequently associated with AATD. Advances in targeted therapies, including augmentation therapy, gene therapy, and RNA-based interventions, have enhanced treatment efficacy and improved patient outcomes. Early detection has also been boosted by the adoption of genetic testing, biomarker assays, and pulmonary function monitoring, enabling timely clinical intervention. Supportive regulatory frameworks, such as orphan drug designations, along with investments in healthcare infrastructure and specialty clinic expansions, are further propelling market growth.
Key Industry Highlights
Augmentation therapy is expected to dominate the therapeutic landscape, accounting for approximately 41% of revenue share in 2026. Gene and RNA-based therapies are projected to be the fastest-growing segment from 2026 to 2033 due to an increasing number of late-stage clinical approvals. In diagnostics, genetic testing is anticipated to lead with an estimated 39% revenue share in 2026, while remote monitoring is forecasted to grow at a CAGR of 14.2%, reflecting rising telehealth adoption. Hospitals are poised to remain the primary end-user with around 46% revenue share in 2026, while specialty clinics and home healthcare are expected to witness the fastest growth, supported by decentralized treatment delivery models. Regionally, North America is projected to dominate with an estimated 42% market share in 2026, whereas Asia Pacific is expected to register the fastest growth at 13.1% CAGR through 2033. Strategic developments in the market include novel delivery technology launches, biotech partnerships, and expansion into emerging economies such as Asia Pacific and Latin America.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/34586
Rising Prevalence Awareness and Enhanced Detection Pathways
The growing incidence of chronic respiratory disorders continues to expand demand for advanced diagnostics and therapies. AATD remains significantly underdiagnosed, prompting the development of comprehensive diagnostic algorithms that integrate serum levels, genotyping, and functional assessments. Efforts by organizations such as the Alpha-1 Foundation, which focus on centralized detection initiatives, outreach, and patient identification, are enhancing diagnosis rates and expanding the addressable patient population. Improved detection is enabling clinicians to initiate earlier and more effective interventions, thus driving therapeutic and diagnostic solution utilization.
Technological Advancements and Regulatory Support
Technological innovations in gene editing, RNA medicine, and next-generation diagnostics are advancing the AATD market. Preclinical and clinical data from novel gene editing candidates demonstrate promising results in correcting SERPINA1 mutations and increasing functional AAT protein levels. Regulatory incentives, including FDA Fast Track and Orphan Drug designations, are facilitating accelerated approval pathways, reducing time-to-market for high-impact therapies. Expanding healthcare infrastructure and regulatory support are enabling high-value diagnostic and therapeutic adoption while lowering barriers to entry for emerging modalities.
Challenges: Treatment Costs and Access Disparities
AATD therapies, especially augmentation and gene-based treatments, involve high costs that can limit patient access, particularly in low- and middle-income countries. Inconsistent reimbursement policies and substantial out-of-pocket expenses restrict therapy adoption and may slow overall market growth. Public health reporting indicates that high-cost treatments for rare diseases often remain inaccessible, even in high-income regions, highlighting systemic barriers to equitable care.
Emerging Market Penetration and Digital Health Integration
Emerging economies, including China, India, and Brazil, are enhancing healthcare infrastructure and specialty care networks, increasing access to advanced AATD interventions. Telehealth adoption and reimbursement expansion enable remote monitoring, patient adherence tracking, and decentralized care delivery. Digital health solutions improve patient engagement, accessibility, and care continuity, opening new service-based revenue opportunities.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/34586
Category-wise Analysis
Genetic testing is projected to hold 39% of market revenue in 2026, supported by widespread clinician adoption and standardized guidelines. Remote monitoring is expected to grow at a CAGR of 14.2%, fueled by telemedicine integration and real-time patient tracking. Hospitals remain the primary care setting, while specialty clinics and home healthcare are rapidly expanding due to patient-centric care models.
Regional Insights
North America is expected to dominate the market with 42% share in 2026, supported by strong infrastructure, insurance coverage, and rare disease care networks. Europe maintains a significant presence with regulatory harmonization and structured reimbursement pathways enhancing adoption. Asia Pacific is projected to grow at the fastest rate due to expanding healthcare infrastructure, rising awareness, and telehealth integration.
Competitive Landscape
The AATD market is moderately consolidated, with leading players such as Grifols, CSL Behring, Takeda, Kamada, and Baxter maintaining strong market positions through clinical networks, R&D investment, and regulatory expertise. Smaller regional and niche players focus on specialized therapies or emerging markets, while digital health innovations enable new entrants to participate through telehealth solutions. Strategic partnerships, acquisitions, and clinical collaborations are expected to drive future market growth.
Market Segmentation
By Therapy
Augmentation Therapy
Gene Therapy
RNA-Based Therapies
Small Molecules
Advanced Biologics
By Diagnostics & Monitoring
Genetic Testing
Biomarker Testing
Pulmonary Function
Imaging
Remote Monitoring
By End-User
Hospitals
Specialty Clinics
Home Healthcare
Diagnostic Centers
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East & Africa
Checkout Now & Download Complete Market Report: https://www.persistencemarketresearch.com/checkout/34586
Key Developments
Recent developments include Beam Therapeutics’ regulatory milestones for gene-editing therapies, Regeneron-Tessera Therapeutics’ US$ 275 million collaboration on gene-writing treatments, and BioMarin’s US$ 4.8 billion acquisition of Amicus Therapeutics, reflecting significant investment in rare genetic disease markets, including AATD.
Read Related Reports:
Atherectomy and Thrombectomy Devices Market: The global atherectomy and thrombectomy devices market will grow from US$4.2B in 2026 to US$6.9B by 2033, registering a steady CAGR of 5.3% during the period.
Spinal Stenosis Implant Market: The spinal stenosis implant market, growing from US$ 10.1 billion in 2026 to US$ 16.6 billion by 2033 at a 5.3% CAGR, driven by surgical innovation.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.